Technology
The Science of Fuel Degradation in Small Engines
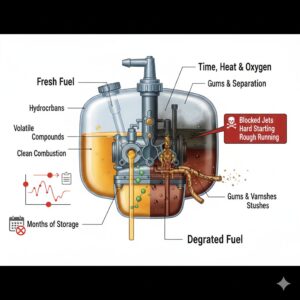
Fuel degradation is one of the least understood yet most damaging processes affecting small garden engines. Lawn mowers, strimmers, chainsaws, and similar machines often fail not because of mechanical wear, but because the fuel they rely on has chemically changed while sitting unused. Understanding what actually happens to fuel over time helps explain many common problems, from hard starting to blocked carburettors and poor performance.
Petrol is not a single substance but a complex blend of hydrocarbons designed to vaporise and burn efficiently. When fresh, these components work together to ignite easily and deliver consistent power. However, petrol is inherently unstable once it leaves the refinery. Exposure to oxygen, heat, and time begins a slow process of chemical breakdown that is especially problematic in small engines.
One of the first stages of fuel degradation is oxidation. As petrol is exposed to air, oxygen reacts with hydrocarbons in the fuel. This reaction alters the chemical structure of the fuel, reducing its volatility. Volatility is critical for starting, particularly in cold conditions, because fuel must vaporise readily to ignite. As oxidation progresses, starting becomes more difficult and combustion less efficient.
At the same time, lighter hydrocarbons begin to evaporate. These lighter fractions are responsible for easy starting and smooth idling. In vented fuel systems, which are common in small engines, evaporation happens continuously. As these components are lost, the remaining fuel becomes heavier and harder to ignite, leading to sluggish starting and rough running.
Ethanol accelerates both oxidation and evaporation. Modern petrol often contains ethanol, which behaves very differently from traditional hydrocarbons. Ethanol is hygroscopic, meaning it absorbs moisture from the surrounding air. Even in sealed containers, temperature changes cause fuel tanks and cans to breathe, allowing moist air to enter and increasing water content in the fuel.
As water accumulates, it introduces a new set of chemical and physical problems. Petrol and water do not mix well, but ethanol acts as a bridge between them. Once the ethanol becomes saturated with water, phase separation can occur. This is the point at which ethanol and water separate from the petrol and settle at the bottom of the tank as a heavier layer.
Phase separation is particularly damaging in small engines. Fuel is usually drawn from the lowest point in the tank, meaning the first fuel delivered to the carburettor after storage may be an ethanol-water mixture with very little usable petrol. This mixture does not burn properly and can cause engines to refuse to start or run erratically.
Another consequence of fuel degradation is the formation of gums and varnishes. As petrol oxidises, it produces sticky residues that cling to internal surfaces. In carburettors, these residues accumulate in jets, float bowls, and fine passages where fuel flow is precisely controlled. Even microscopic deposits can disrupt the air-fuel mixture enough to prevent starting or cause surging and stalling.
Small engines are especially vulnerable because their carburettors are simple and finely calibrated. Unlike modern car engines, they lack sensors and electronic controls that can compensate for poor fuel quality. Any restriction or imbalance in fuel delivery is immediately felt in performance.
Materials inside fuel systems also play a role. Ethanol acts as a solvent, gradually breaking down rubber hoses, seals, and gaskets. As these components degrade, they may release particles into the fuel, further contaminating it. Hoses can also soften or collapse internally, restricting fuel flow and compounding starting problems caused by degraded fuel.
Temperature fluctuations worsen degradation. Heat accelerates chemical reactions, increasing oxidation rates, while cooling encourages condensation. In sheds and garages, daily temperature swings repeatedly warm and cool fuel systems. This cycling speeds up evaporation, moisture absorption, and residue formation, particularly when equipment is stored with partially filled tanks.
Time is the final critical factor. Fresh petrol may perform adequately for a few weeks, but degradation accelerates as storage length increases. After several months, particularly with ethanol-blended fuel, petrol can become unusable. In small engines that are used seasonally, this timescale aligns perfectly with common storage periods, making fuel degradation a predictable cause of failure.
Two-stroke engines face additional challenges. These engines rely on petrol mixed with oil for lubrication. Over time, this mixture can separate, especially in the presence of ethanol. If fuel is not mixed evenly before use, lubrication becomes inconsistent, increasing wear and raising operating temperatures. Degraded fuel in two-stroke engines therefore affects both combustion and lubrication.
The science behind long-life and alkylate fuels illustrates how these problems can be reduced. These fuels are refined to remove unstable hydrocarbons and contain no ethanol. As a result, they resist oxidation, do not absorb moisture, and produce far fewer residues. Their chemical stability explains why they can remain usable for years rather than months.
Service professionals regularly see the practical effects of fuel chemistry. Dealers such as Ron Smith often encounter machines that appear to have serious mechanical faults but are actually suffering from predictable chemical degradation of fuel. Once fuel systems are cleaned and more stable fuel is used, many of these machines return to normal operation.
Fuel stabilisers attempt to slow degradation by inhibiting oxidation and evaporation. While they can extend fuel life, they do not stop degradation entirely. Their effectiveness depends on being added to fresh fuel and circulated through the entire system before storage. They cannot reverse chemical changes once fuel has already broken down.
Understanding fuel degradation also explains why topping up old fuel rarely works. Adding fresh petrol to degraded fuel simply dilutes the problem without eliminating it. Oxidised compounds and moisture remain present, continuing to affect performance and accelerate further breakdown.
Prevention is therefore more effective than cure. Using fresh fuel, avoiding long storage periods with petrol left in tanks, and choosing fuels with greater chemical stability all reduce degradation. Storage practices such as keeping containers sealed, tanks full, and fuel cool also slow the underlying chemical processes.
From a scientific perspective, fuel degradation is inevitable. Petrol is not designed for long-term storage, and small engines are not designed to tolerate degraded fuel. Problems arise not because owners do something unusual, but because normal usage patterns clash with fuel chemistry.
Recognising this mismatch changes how fuel is viewed. Rather than being a simple consumable, fuel becomes a time-sensitive component of engine health. When fuel quality aligns with how and how often machines are used, reliability improves dramatically.
In practical terms, understanding the science behind fuel degradation allows owners to make informed decisions. Choosing the right fuel, managing storage carefully, and respecting the chemical limits of petrol all help small engines perform as intended. When fuel is treated as a critical system rather than an afterthought, many of the most common and costly garden machinery problems simply never occur.