Technology
How Laser Engraving Drives Medical Device Traceability in Australia
In Australia’s high-stakes medical technology sector—ranging from the biotech hubs of Melbourne to innovative surgical startups in Sydney—quality is non-negotiable. At the heart of this industry lies a critical requirement: traceability.
With the Therapeutic Goods Administration (TGA) continuously aligning Australian regulations with global standards, the permanent identification of medical instruments has moved from a “best practice” to a strict legal necessity. Laser engraving has emerged as the premier technology to meet these rigorous demands.
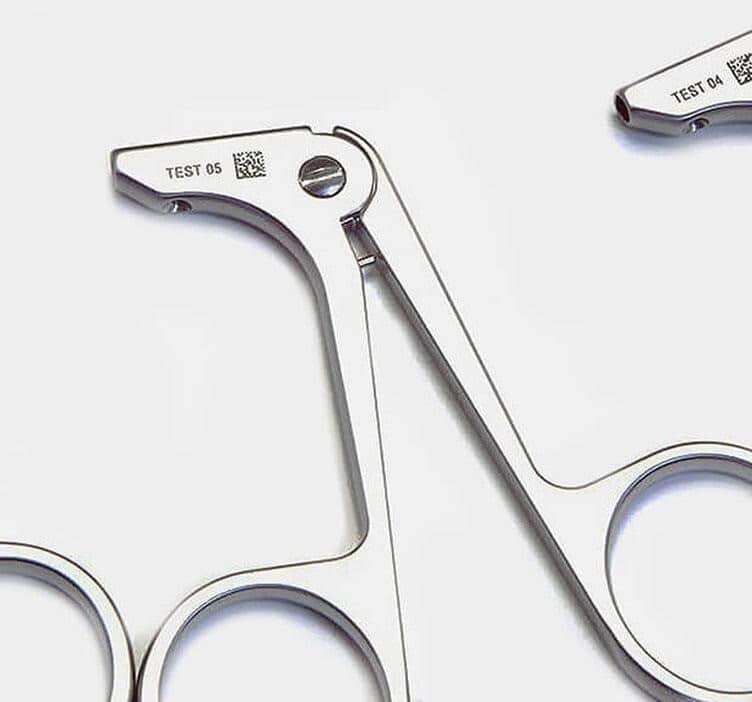
1. The Regulatory Landscape: UDI and the TGA
To enhance patient safety, the Australian government and the TGA have moved toward a mandatory Unique Device Identification (UDI) system. This requires medical devices to carry a unique identifier that is both human-readable and machine-readable (typically a 2D Data Matrix code).
Why Laser Engraving is the Standard:
Unlike adhesive labels or ink-based markings—which can peel, fade, or harbour bacteria—a laser mark is:
- Permanent: It lasts the entire functional life of the instrument.
- Indelible: It remains perfectly legible even after thousands of high-pressure autoclave (sterilisation) cycles.
- Tamper-Proof: The mark is etched directly into the material, preventing accidental or intentional removal.
2. Technical Advantages for Aussie Manufacturers
Australian medical manufacturers face unique challenges, from strictly regulated export requirements to the need for extreme durability in diverse clinical environments. Laser marking offers specific technical benefits:
Hygiene and Surface Integrity
For surgical tools, the marking process must not compromise the “passivation layer” of the metal, as this prevents rust. Laser technology allows for Annealing Markings. Instead of removing material, the laser uses controlled heat to create an oxidation layer beneath the surface. The result is a smooth, dark mark that is flush with the surface, leaving no crevices for bacteria to grow.
Precision on Micro-Scales
Whether it is marking micro-implants for spinal surgery or fine dental instruments, lasers can produce high-density codes on incredibly small surface areas without causing structural stress to the device.
Resistance to Harsh Chemicals
Medical devices in Australian hospitals undergo rigorous cleaning with aggressive chemical disinfectants. Laser marks are chemically inert and will not react or degrade, ensuring the device remains identifiable from the operating theatre to the sterile processing department.

3. Selecting the Right Laser Source
In the medical field, two laser types dominate the production line:
- Fibre Lasers: The industry workhorse for metals such as stainless steel, titanium, and cobalt-chrome. This is the go-to for surgical instruments and orthopaedic implants.
- UV Lasers: Known as “Cold Marking” lasers. These are essential for heat-sensitive plastics, catheters, and thin-walled tubing, as they mark the surface without creating a thermal “heat-affected zone” that could weaken the material.
4. Integration and “Industry 4.0”
For Australian manufacturers looking to remain competitive on the global stage, integrating laser marking into a digital workflow is key:
- ERP Connectivity: Modern laser systems connect directly to your database, automatically generating serial numbers and batch data to eliminate human error.
- Automated Verification: Integrated vision systems can scan the mark immediately after engraving, “grading” the code to ensure it meets international readability standards before it leaves the factory.
5. Why Choose Laser for Traceability?
| The Challenge | The Laser Solution |
| Regulatory Compliance | Full TGA and UDI alignment with permanent codes. |
| Sterilisation | Withstands repeated autoclaving without fading. |
| Infection Control | Smooth, non-porous marks prevent bacterial growth. |
| Material Diversity | High-contrast results on titanium, steel, and polymers. |
Laser engraving is far more than just a branding tool; it is a vital safety feature. For Australian medical device manufacturers, investing in high-quality laser marking technology is an investment in brand reputation, legal security, and—most importantly—patient outcomes.
At OMTech Australia, we provide advanced Fibre and UV laser solutions designed to meet the exacting standards of the medical industry. We help you ensure that every device you produce is 100% traceable, 100% of the time.



















